INTENSIVE LDL CHOLESTEROL TARGET IMPROVES OUTCOMES IN CARDIOVASCULAR DISEASE
The Ez-PAVE trial randomized patients with cardiovascular disease to LDL targets of less than 55 mg/dL versus less than 70 mg/dL
Straight Healthcare
April 2026
April 2026
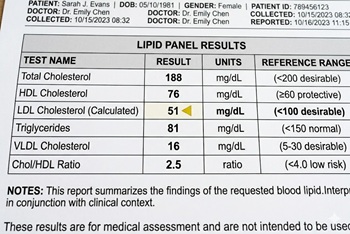
Recent American Heart Association (AHA) cholesterol treatment guidelines emphasize aggressive lipid management for secondary prevention in patients with atherosclerotic cardiovascular disease. For years, the recommended low-density lipoprotein (LDL) cholesterol target has been less than 70 mg/dl. More recently, guidelines have introduced an optional, stricter target of less than 55 mg/dl for patients deemed to be at very high risk for recurrent cardiovascular events. However, recommendations for the lower target have been based largely on post-hoc analyses and trials evaluating the addition of other therapies (such as ezetimibe or PCSK9 inhibitors) to statins, not the targets themselves. Consequently, evidence from randomized trials supporting an LDL target below 55 mg/dL has been lacking.
To address this clinical gap, researchers conducted the Ez-PAVE trial, an open-label, randomized study that enrolled 3,048 South Korean patients with documented atherosclerotic cardiovascular disease (average age 64 years; median baseline LDL 76 mg/dl). Participants were randomly assigned in a 1:1 ratio to a target LDL cholesterol level of less than 55 mg/dL (intensive group) or less than 70 mg/dL (conventional group). Adjustments to lipid-lowering therapies, including high-intensity statins, ezetimibe, and PCSK9 inhibitors, were made at the discretion of treating physicians. Over a median follow-up of 3 years, the median LDL cholesterol level achieved was 56 mg/dl in the intensive group and 66 mg/dl in the conventional group. The primary endpoint (a composite of death from cardiovascular causes, nonfatal myocardial infarction, nonfatal stroke, any revascularization, or hospitalization for unstable angina) occurred in 6.6% of patients in the intensive group and 9.7% of patients in the conventional group (Hazard Ratio [HR]: 0.67; 95% CI, 0.52 to 0.86; P=0.002). Prespecified safety endpoints, such as new-onset diabetes and statin-associated muscle symptoms, were similar between groups, with the intensive group notably showing a lower incidence of clinically significant creatinine elevation.
The Ez-PAVE trial provides direct evidence supporting the "lower is better" hypothesis for cholesterol in secondary prevention. Unlike prior trials that validated specific lipid-lowering medications, Ez-PAVE isolated the benefit of the LDL target itself. These findings support current recommendations and affirm that an LDL below 55 mg/dL should be the goal in most patients with established atherosclerotic cardiovascular disease. Additionally, concerns over worse side effects with more intensive therapy were not borne out in this study. One weakness of the study was that it was conducted exclusively in South Korea, thereby lacking ethnic diversity.
To address this clinical gap, researchers conducted the Ez-PAVE trial, an open-label, randomized study that enrolled 3,048 South Korean patients with documented atherosclerotic cardiovascular disease (average age 64 years; median baseline LDL 76 mg/dl). Participants were randomly assigned in a 1:1 ratio to a target LDL cholesterol level of less than 55 mg/dL (intensive group) or less than 70 mg/dL (conventional group). Adjustments to lipid-lowering therapies, including high-intensity statins, ezetimibe, and PCSK9 inhibitors, were made at the discretion of treating physicians. Over a median follow-up of 3 years, the median LDL cholesterol level achieved was 56 mg/dl in the intensive group and 66 mg/dl in the conventional group. The primary endpoint (a composite of death from cardiovascular causes, nonfatal myocardial infarction, nonfatal stroke, any revascularization, or hospitalization for unstable angina) occurred in 6.6% of patients in the intensive group and 9.7% of patients in the conventional group (Hazard Ratio [HR]: 0.67; 95% CI, 0.52 to 0.86; P=0.002). Prespecified safety endpoints, such as new-onset diabetes and statin-associated muscle symptoms, were similar between groups, with the intensive group notably showing a lower incidence of clinically significant creatinine elevation.
The Ez-PAVE trial provides direct evidence supporting the "lower is better" hypothesis for cholesterol in secondary prevention. Unlike prior trials that validated specific lipid-lowering medications, Ez-PAVE isolated the benefit of the LDL target itself. These findings support current recommendations and affirm that an LDL below 55 mg/dL should be the goal in most patients with established atherosclerotic cardiovascular disease. Additionally, concerns over worse side effects with more intensive therapy were not borne out in this study. One weakness of the study was that it was conducted exclusively in South Korea, thereby lacking ethnic diversity.
- Intensive LDL Cholesterol Targeting in Atherosclerotic Cardiovascular Disease, NEJM (2026) [PubMed abstract]
- Cholesterol treatment guidelines
